 Safe handling techniques enable the transport liquid fluorine by the ton. The recommended maximum allowable concentration for a daily 8-hour time-weighted exposure is 1 ppm. Iridium is known for being the rarest element and has numerous applications in diverse industries. The free element has a characteristic pungent odor, detectable in concentrations as low as 20 ppb, which is below the safe working level. HandlingĮlemental fluorine and the fluoride ion are highly toxic. Compounds of fluorine with rare gases have now been confirmed in fluorides of xenon, radon, and krypton. (a) Which of these have physical and chemical properties. One hypothesis says that fluorine can be substituted for hydrogen wherever it occurs in organic compounds, which could lead to an astronomical number of new fluorine compounds. Chorine in the Periodic Table is surrounded by the elements with atomic number 9, 16, 18 and 35. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and. The presence of fluorine as a soluble fluoride in drinking water to the extent of 2 ppm may cause mottled enamel in teeth when used by children acquiring permanent teeth in smaller amounts, however, fluoride helps prevent dental cavities.Įlemental fluorine has been studied as a rocket propellant as it has an exceptionally high specific impulse value. Element Fluorine (F), Group 17, Atomic Number 9, p-block, Mass 18.998. Fluorochlorohydrocarbons are extensively used in air conditioning and refrigeration. Hydrofluoric acid etches glass of light bulbs. Usesįluorine and its compounds are used in producing uranium (from the hexafluoride) and more than 100 commercial fluorochemicals, including many high-temperature plastics. The nuclear bomb project and nuclear energy applications, however, made it necessary to produce large quantities. Until World War II, there was no commercial production of elemental fluorine. Finely divided metals, glass, ceramics, carbon, and even water burn in fluorine with a bright flame. It is a pale yellow, corrosive gas, which reacts with most organic and inorganic substances. Propertiesįluorine is the most electronegative and reactive of all elements. The element was finally isolated in 1866 by Moissan after nearly 74 years of continuous effort. Scheele and many later investigators, including Davy, Gay-Lussac, Lavoisier, and Thenard, experimented with hydrofluoric acid, some experiments ending tragically. 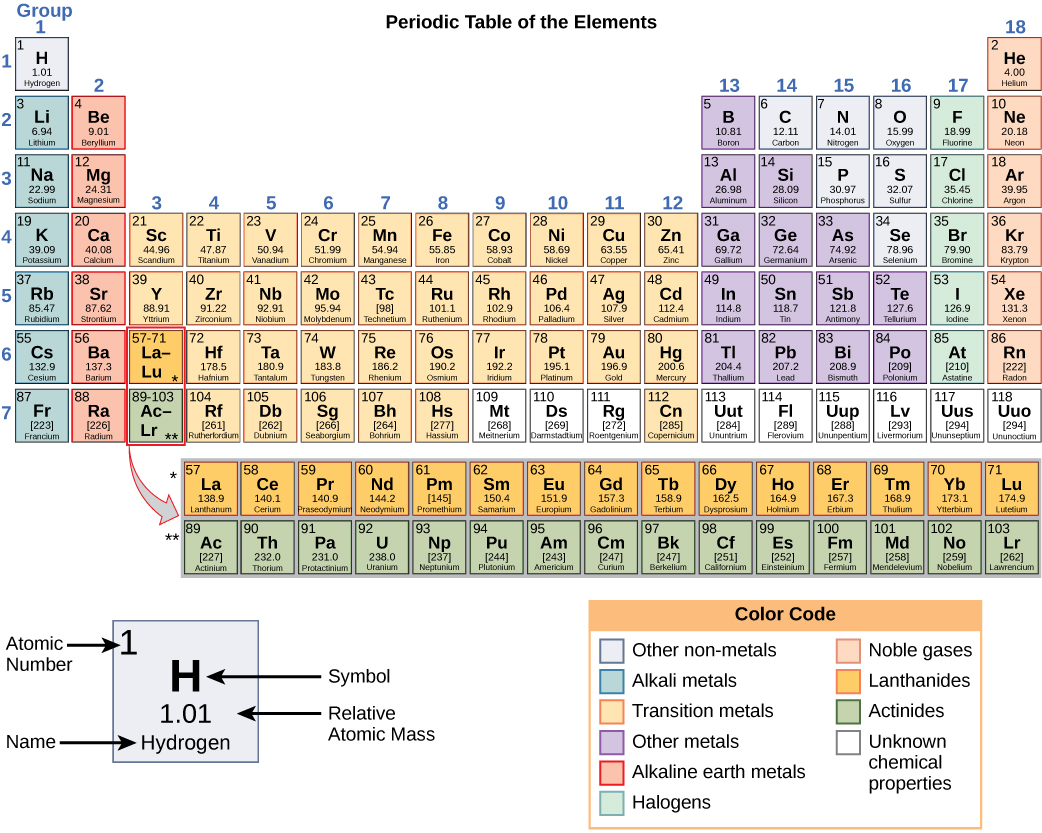  
In 1529, Georigius Agricola described the use of fluorspar as a flux, and as early as 1670 Schwandhard found that glass was etched when exposed to fluorspar treated with acid. Fluorine is found in nature in the form of calcium fluoride, CaF 2, called fluorite, which forms regular crystals.įrom the Latin and French fluere: flow or flux. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
