 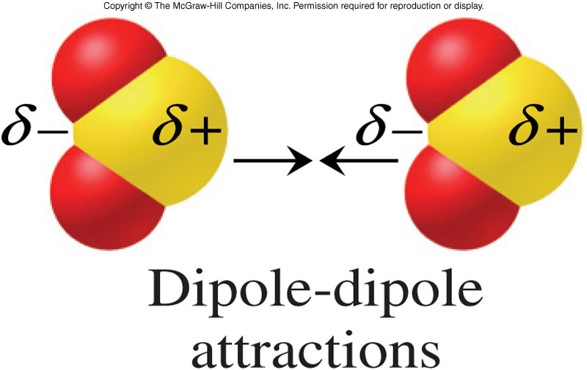
Conversely, if measurements show that a molecular dipole is zero, it gives an indication of the symmetry of the molecule. Symmetry arguments can explain the vanishing of components of a molecular dipole (symmetry gives a " selection rule"), but symmetry arguments cannot directly predict the magnitudes of the non-vanishing components. Whether a molecule has one or more non-zero permanent dipole components depends on the symmetry of the molecule and on the symmetry species of the molecular state under consideration. Dipole moments can be computed reliably for smaller molecules (say up to 10 second-period atoms) by means of quantum chemical methods. Molecular dipole moments may be obtained experimentally the main techniques are microwave spectroscopy and measurements of dielectric constants as function of temperature. The figure shows the dipole moment of water in the physics convention. For physicists the direction of the dipole vector is from negative to positive charge and for many chemists it is the opposite, namely from positive to negative charge. However, all scales predict the same direction of the dipole. These concepts are semi-quantitative and different scales for electronegativity and electropositivity of the chemical elements are in use, leading to differing values of molecular dipole moments. An electronegative atom attracts electrons (becomes negative) and an electropositive atom donates electrons (becomes positive). In chemistry, polarity is usually explained by the presence of electronegative and/or electropositive atoms in the molecule. The size of this dipole, referred to as the molecular dipole moment, gives an indication of the polarity of the molecule, that is, the dipole moment is a measure for the amount of charge separation in a molecule. Because of symmetry the vector has one non-vanishing component it points from the negative to the positive region of the molecule.īeing a charge distribution, a molecule may possess a permanent electric dipole, a vector known as the molecular dipole. The molecule is electrically neutral: 2δ + + δ 2 − = 0. It also has a great deal to do with the function of water as the solvent of life in biological systems.Fig. This can lead to condensation and phenomena like cloud formation, fog, the dewpoint, etc. It causes water vapor at sufficient vapor pressure to depart from the ideal gas law because of dipole-dipole attractions. The polar nature of the water molecule has many implications. The polar nature of water molecules allows them to bond to each other in groups and is associated with the high surface tension of water. Microwaves can add energy to the water molecules, whereas molecules with no dipole moment would be unaffected. The dipole moment of water provides a "handle" for interaction with microwave electric fields in a microwave oven. This dipolar interaction is an example of hydrogen bonding. It also contributes to the fact that water has an unusually high boiling point. The dipolar interaction between water molecules represents a large amount of internal energy and is a factor in water's large specific heat. 15 nm for the effective radius of hydrogen in liquid form, so the charge separation is small compared to an atomic radius. 05 nm for the first Bohr radius of a hydrogen atom and about. Treating this system like a negative charge of10 electrons and a positive charge of 10e, the effective separation of the negative and positivecharge centers is The measured magnitude of this dipole moment is The asymmetry of the water molecule leads toa dipole moment in the symmetry plane pointed toward the more positive hydrogen atoms. HyperPhysics***** Electricity and Magnetism This is called polarization and the magnitude of the dipole moment induced is a measure of the polarizability of the molecular species. Even if there is no permanent dipole moment, it is possible to induce a dipole moment by the application of an external electric field. Molecules with mirror symmetry like oxygen, nitrogen, carbon dioxide, and carbon tetrachloride have no permanent dipole moments. A good example is the dipole moment of the water molecule. 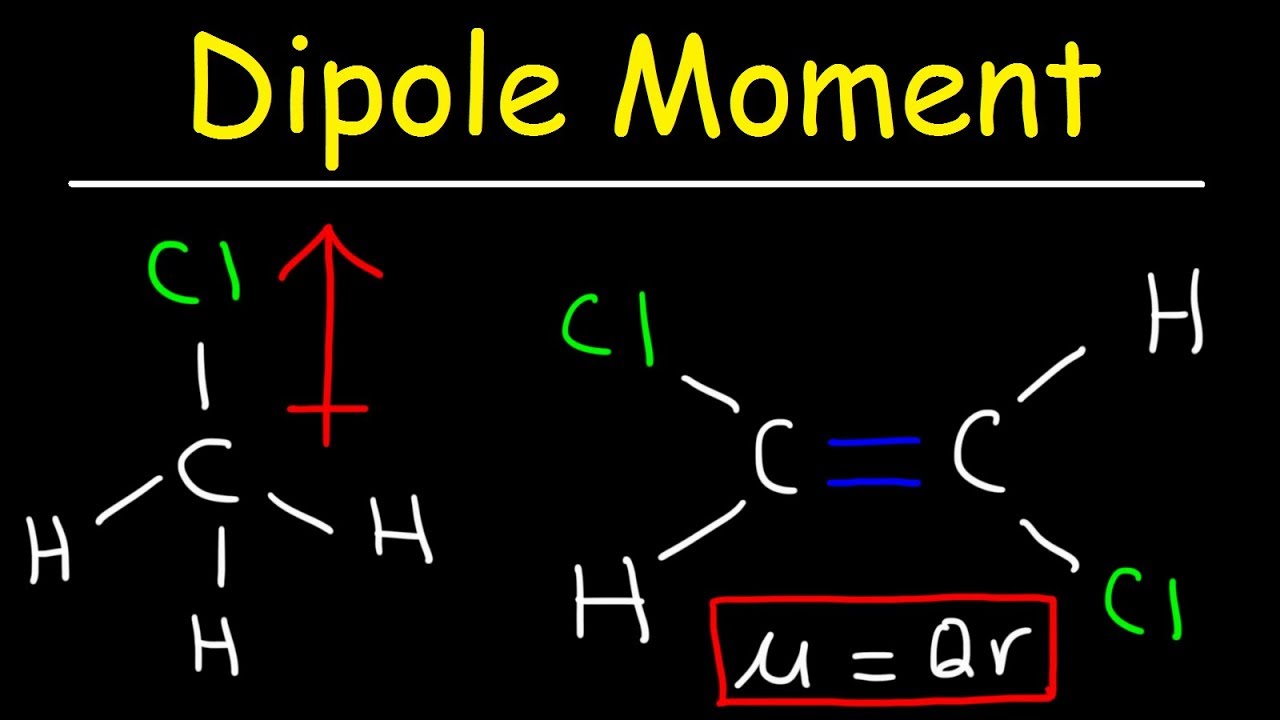
Such molecules are said to be polar because they possess a permanent dipole moment. Molecular Dipole Moments Molecular Dipole MomentsĮven though the total charge on a molecule is zero, the nature of chemical bonds is such that the positive and negative charges do not completely overlap in most molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
